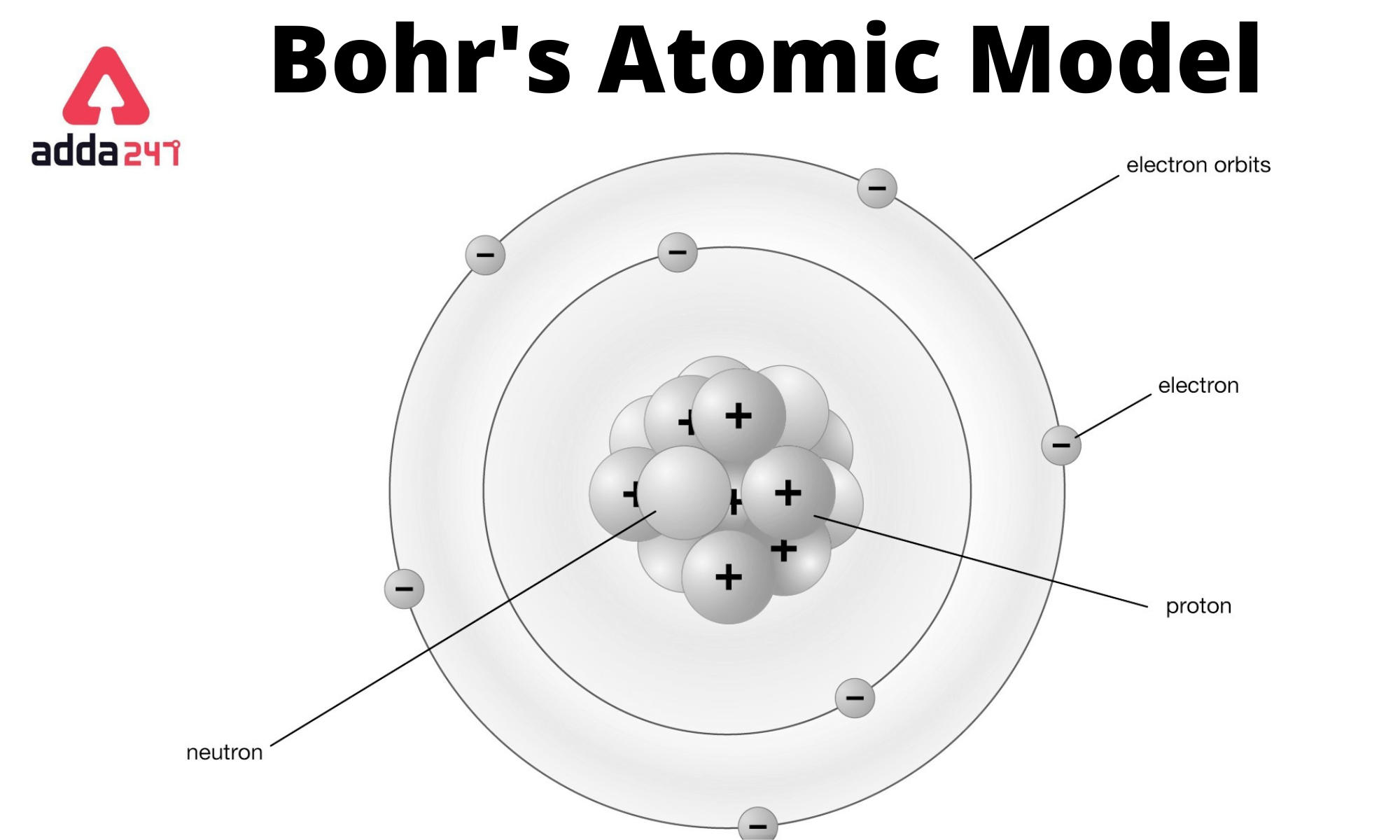  
Atoms have protons and neutrons in the center, making the nucleus, while the electrons orbit the nucleus. Protons carry a positive electrical charge. The three parts of an atom are protons, neutrons, and electrons. They do consist of smaller parts, but can only be broken by nuclear reactions. In contrast, the particles that were highly deflected must have experienced a tremendously powerful force within the atom.\): The Structure of the Atom. Atoms are the smallest unit of matter that cannot be divided using any chemical method. Some said everything was made of water, which comes in three forms (solid ice, liquid water, and gaseous steam). Beginning in about 600 BC, many Greek philosophers struggled to understand the nature of matter. 3.1: Atomic Theory is shared under a not declared license and was authored, remixed. Atomic theory is the description of atoms, the smallest units of elements. As a theory, it can be derived as a first-order approximation of the hydrogen atom using the broader and much more accurate quantum mechanics and thus may be considered to be an obsolete scientific theory. Each element has its own atomic number, which is equal to the number of protons in its nucleus. The Bohr model is a relatively primitive model of the hydrogen atom, compared to the valence shell model. Atoms themselves are composed of protons, neutrons, and electrons. Because the vast majority of the alpha particles had passed through the gold, he reasoned that most of the atom was empty space. atomic model, in physics, a model used to describe the structure and makeup of an atom. Britannica Quiz Matter and More Quiz How does Niels Bohrs atomic model work An overview of Niels Bohrs refinement of the Rutherford model. Chemistry is based on the modern atomic theory, which states that all matter is composed of atoms. Much of Lavoisier’s work as a chemist was. Rutherford needed to come up with an entirely new model of the atom in order to explain his results. The development of the atomic theory owes much to the work of two men: Antoine Lavoisier, who did not himself think of matter in terms of atoms but whose work laid organization groundwork for thinking about elements, and John Dalton, to whom the atomic theory is attributed. In a famous quote, Rutherford exclaimed that it was "as if you had fired a 15-inch shell at a piece of tissue and it came back and hit you." Dalton based his theory on the law of conservation of mass and the law of constant composition. No prior knowledge had prepared them for this discovery. Daltons atomic theory was the first complete attempt to describe all matter in terms of atoms and their properties. Some were even redirected back toward the source. Surprisingly, while most of the alpha particles were indeed not deflected, a very small percentage (about 1 in 8000 particles) bounced off the gold foil at very large angles. Rutherford found that a small percentage of alpha particles were deflected at large angles, which could be explained by an atom with a very small, dense, positively-charged nucleus at its center (bottom).Īccording to the accepted atomic model, in which an atom's mass and charge are uniformly distributed throughout the atom, the scientists expected that all of the alpha particles would pass through the gold foil with only a slight deflection or none at all. Thomson conducted some research that suggested that Daltons Atomic Theory was not the. In fact, for almost 100 years, it seemed as if Daltons Atomic Theory was the whole truth. For example, if an element such as copper consists of only one kind of atom, then it cannot be broken down into simpler substances, that is, into substances composed of fewer types of atoms. This is a list of the basic characteristics of atoms: Atoms cannot be divided using chemicals. Daltons Atomic Theory held up well to a lot of the different chemical experiments that scientists performed to test it. One hundred years ago, Niels Bohrs pioneering paper on the electronic structure of the hydrogen atom revolutionized atomic theory. (B) According to the plum pudding model (top), all of the alpha particles should have passed through the gold foil with little or no deflection. Dalton’s atomic theory provides a microscopic explanation of the many macroscopic properties of matter that you’ve learned about. \): (A) The experimental setup for Rutherford's gold foil experiment: A radioactive element that emitted alpha particles was directed toward a thin sheet of gold foil that was surrounded by a screen which would allow detection of the deflected particles.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
